 
The half reactions, balanced with acid, are:Īt the positively charged anode, an oxidation reaction occurs, generating oxygen gas and giving electrons to the anode to complete the circuit: In pure water at the negatively charged cathode, a reduction reaction takes place, with electrons (e −) from the cathode given to hydrogen cations to form hydrogen gas. Efficiency is increased through the addition of an electrolyte (such as a salt, an acid or a base) and electrocatalysts.ĭiagram showing the overall chemical equation. Many electrolytic cells lack requisite electrocatalysts. Pure water has an electrical conductivity about one-millionth that of seawater. This is in part due to the limited self-ionization of water. Without the excess energy, electrolysis occurs slowly or not at all. 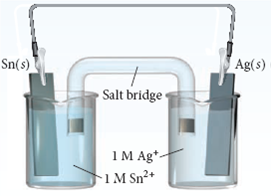
However, in many cells competing side reactions occur, resulting in additional products and less than ideal faradaic efficiency.Įlectrolysis of pure water requires excess energy in the form of overpotential to overcome various activation barriers. Assuming ideal faradaic efficiency, the amount of hydrogen generated is twice the amount of oxygen, and both are proportional to the total electrical charge conducted by the solution. 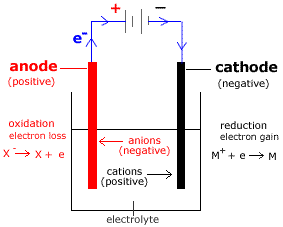
Hydrogen appears at the cathode (where electrons enter the water), and oxygen at the anode. 
Principles Ī DC electrical power source is connected to two electrodes, or two plates (typically made from an inert metal such as platinum or iridium) that are placed in the water. A method of industrial synthesis of hydrogen and oxygen through electrolysis was developed by Dmitry Lachinov in 1888. Zénobe Gramme invented the Gramme machine in 1869, making electrolysis a cheap method for hydrogen production. He used a high voltage battery and non-reactive electrodes and vessels such as gold electrode cones that doubled as vessels bridged by damp asbestos. In 1806 Humphry Davy reported the results of extensive distilled water electrolysis experiments, concluding that nitric acid was produced at the anode from dissolved atmospheric nitrogen. In 1800 Alessandro Volta invented the voltaic pile, while a few weeks later English scientists William Nicholson and Anthony Carlisle used it to electrolyse water. In 1789, Jan Rudolph Deiman and Adriaan Paets van Troostwijk used an electrostatic machine to make electricity that was discharged on gold electrodes in a Leyden jar. #color(blue)(2"Ag"^(+)(aq) + "SO"_2(g) + 2"H"_2"O"(l) -> 2"Ag"(s) + "SO"_4^(2-)(aq) + 4"H"^(+)(aq))#įor this reaction, we then = + E_(o "0.80 V" + (-"0.20 V") = color(blue)(+"0.60 V")#Īnd we do not multiply or #E_(o by any coefficients since the scaled mols of electrons and scaled mols of reactants/products cancel out.Device invented by Johann Wilhelm Ritter to develop the electrolysis of water Thus, we reverse the other half-reaction to write it as an oxidation and the decomposition of sulfurous acid leads to the oxidation of sulfur dioxide to sulfate at the anode. The silver reduction has a more positive so reduction is more spontaneous for #"Ag"^+# and #"Ag"^(+)# is reduced at the cathode. Ignoring the charge balance, the main action going on in one half-reaction is: 
0 Comments
Leave a Reply. |

 RSS Feed
RSS Feed
